States of Matter

Atom, Chemical Bond, Chemical Change, Chemical Formula, Chemical Properties, Covalent Bond, Dot Diagram, Double Bond, Element, Energy Levels, Group, Ion, Ionic Bond, Matter, Molecule, Nonpolar Bond, Period, Periodic Table, Polar Bond, Physical Change, Physical Properties, Scientific Theory, Triple Bond, Valence Electrons
Students are to create a list and print the "glossary", stapling it in their notebook. This is a large assignment and should be completed over time
Students are to create a list and print the "glossary", stapling it in their notebook. This is a large assignment and should be completed over time
Identify Physical & Chemical Properties |
Read Chemical Interactions -
"Observing Chemical Change", pp. 46-53 Chemical and Physical Change Lab (Online Activity) |
Making Sense of Density |
Problem:
Is density a characteristic property of a substance? Materials: Balance Water Paper Towels Wooden Stick (app. 6 cm long) Ball of clay (app. 5 cm wide) Crayon without paper Graduated Cylinder, 100 ml Procedure: Follow Steps on p 21 in Chemical Building Blocks Data Table: Copy Data Table Analyze & Conclude: Label the next full page in your notebook "Making Sense fo Density". Answer questions on separate loose-leaf paper. When corrected paper is returned, staple on this page.
| ||||||
Chemical & Physical Change |
Change what's the difference?
Chemical and Physical |
Test Study Guide |
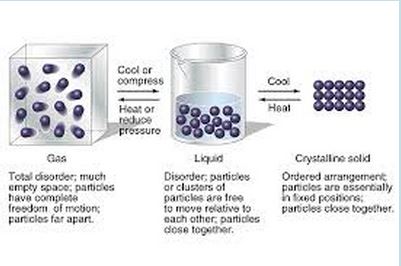
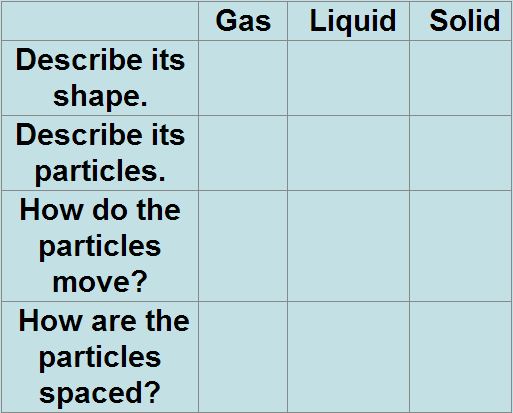
Be able to describe the movement and behavior of particles in solids, liquids, and gases
Explain how matter changes states between solids, liquids, gases Know what density is and how to calculate it Identify and distinguish between physical and chemical characteristics of matter Identify chemical change Identify physical change Recall: "Making Sense of Density" & "Where's the Evidence" Draw and identify the behavior of particles at each state of matter - This is very important it is 1/3 of the test, see chart above |
Making a Model (Solo Cup) |
Question:
What makes up matter? Data & Observations: Draw what items you believe are inside the cup/box, leave 5-8 lines Conclusion: Compare your data and observations with the actual items. Were you correct? |
DOT Diagram |
Read Chemical Interactions - "Atoms, Bonding, and the Periodic Table", pp12-20
What is a dot diagram? What does it tell us? VIDEO ON VALENCE ELECTRIONS Watch video, but only listen to what valence elections are and how to find the number of valence electrons. We will create a simplified (general) Lewis Dot Diagram. Choose any element and draw DOT diagrams. Using the "Blank Periodic Table" (1) neatly print each element symbol in the appropriate cell. Show the number of valence electrons using dots as discussed in class. (2) Show where periods 6 and 7 belong. (3) Using different colored highlighters or lightly shading with colored pencils shade the different types of elements (metals, nonmetals...). (4) Using a different colored thin markers, distinguish between states of matter. (5) Create a key. [3 Ways to Find V.E. - We simplified Groups 3-10]
|
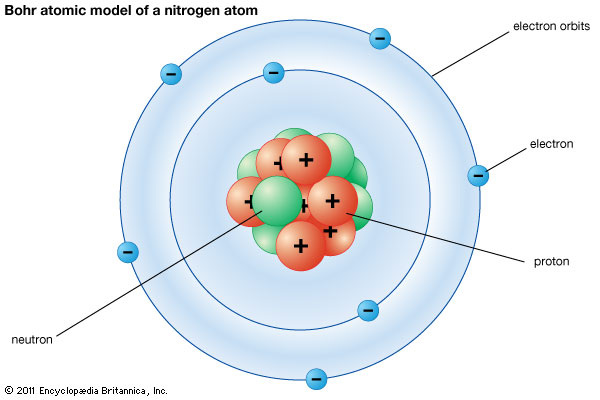
Energy Levels (Tennis Balls) |
Question:
How do valence electrons work in an atom and in bonding? Analysis & Conclusions: Skip lines to answer questions. Explain what each tennis ball represents? How many electrons must be in the outer energy level foran atom to be stable? Describe the two ways atoms combine/bond? Define ionic bond. Define covalent bond. |
Atomic Bonds |
Read Chemical Interactions - "Ionic Bonding", pp. 22-27
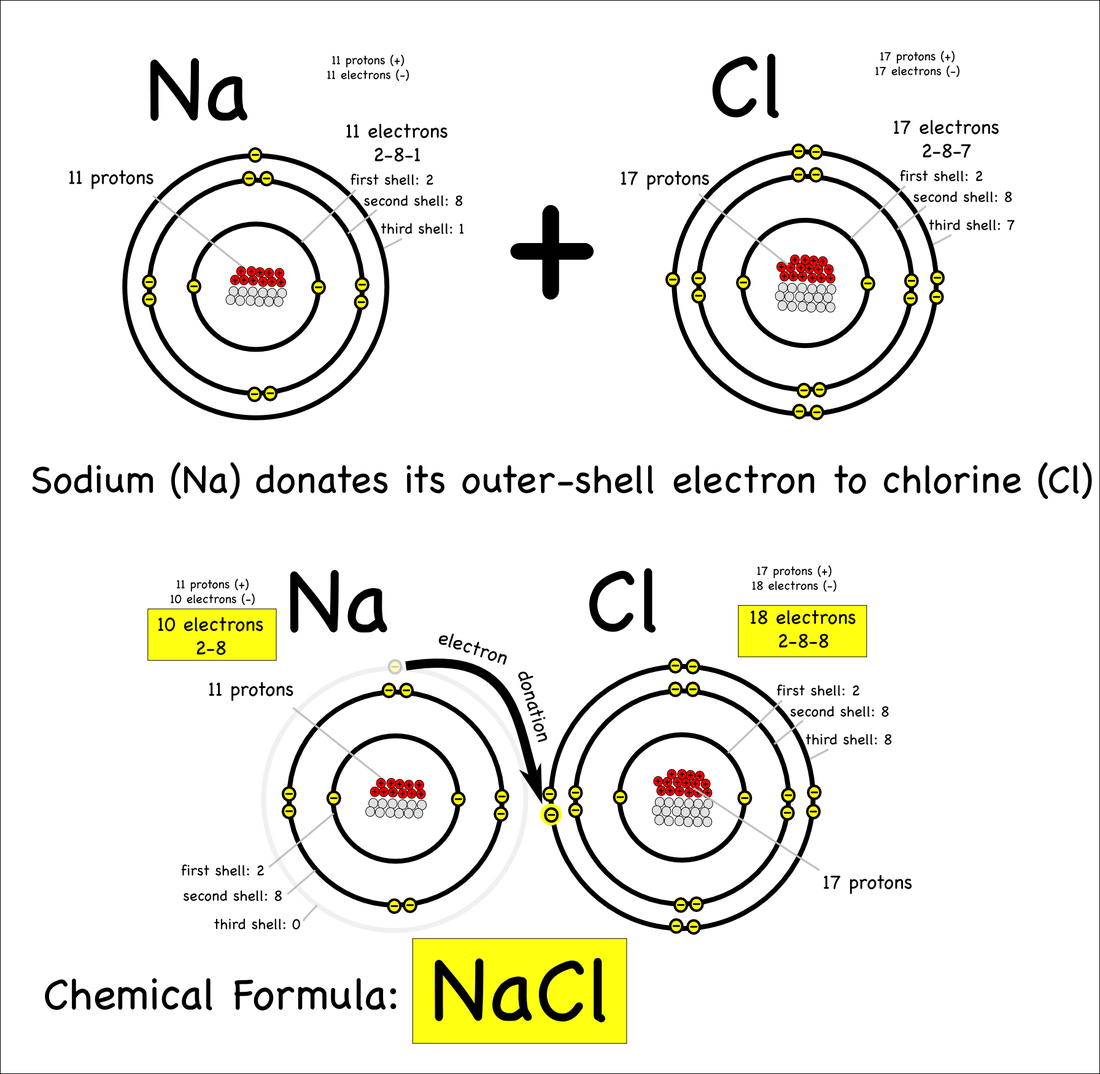
Copy in a notebook, skip lines to answer questions. What do red and black checks represent? Explain how ions form? (Checkers) What happens when a red checker is added? Subtracted? Terms to know: poly, dio, mono, -ide, -ate, -ite See pages 22 and 25 Predict How Ions will bond, write your answers. (Textbook)
EX: Ca 2+ and O 2- will combine to make CaO2 |
Shedding Light on Ions |
Copy the following information in the note section of your your science notebook form pp. 28-29. Place the experiment title in the margin. Be neat. To prepare for this experiment, copy the "Question" and "Data Table" in your notebook Question: Do ionic compounds have an electrical charge? Data Table: Modified from textbook Title one the next full page for this section. You will complete this assignment, turn it in, and staple the graded copy here when it is returned.
Analyze & Conclude: Questions 3, 4, & 5 ONLY |
Candy Compounds |
To prepare for this experiment, copy the "Question" and "Data Table" in your notebook Question: How to determine the type of compound, diagram electron bonding, and write ionic formulas and covalent bonds. Points to Remember: Ions have a charge, their attraction is based on a + and - attraction, both must be balanced Ionic compounds are formed between metals and nonmetals In forming an ionic bond the ion with more V.E.'s takes an electron giving it a - charge Covalent compounds are usually formed between two nonmetals
|
Test Studyguide |
Prefixes: "mono" and "dio"
Know chemical symbols for common elements like: Carbon, Oxygen, Nitrogen, Iron, Sodium Know how to read the Periodic Table (atomic number, atomic mass, know how to find protons, valence electrons, the difference between groups and periods, and type of matter) Scientific Theory Valence Electrons Be able to diagram an atom Review the "Shoebox/Solo Cup" mini lab Be able to determine the atoms of an element in a chemical formula Be able to calculate Atomic Mass Know what matter is Know what an atom, element, and molecules are Be able to read a Dot Diagram Know the difference between an ionic and covalent compounds and bonds Diagram ionic and covalent compounds and bonds |
Extra Credit |
These "gizmos" may not work for all people.
Gizmo Lessons in Chemistry - Print the lesson and complete it online Gizmo Lessons on Properties of Matter - Print the lesson and complete it online |